
By Beata Mostafavi | Michigan Medicine Health Lab | 22 January
 Dr. Venetti (right) with Postdoctoral Fellow Chan Chung, PhD (left)The prognosis for all children diagnosed with an aggressive brain tumor known as diffuse intrinsic pontine glioma (DIPG) and similar tumors has been mostly the same: dismal.
Dr. Venetti (right) with Postdoctoral Fellow Chan Chung, PhD (left)The prognosis for all children diagnosed with an aggressive brain tumor known as diffuse intrinsic pontine glioma (DIPG) and similar tumors has been mostly the same: dismal.
But a small subset of patients with these tumors that bear mutations in a gene in the basic packaging of DNA (known as histone mutations) may have better outcomes than others, suggests new research from Michigan Medicine’s Pediatric Brain Tumor Research Initiative.
Researchers mined data from more than 500 published cases of tumors with these histone mutations across the globe between 2012 to 2017 — including Chad Carr’s tumor. Chad, the grandson of former University of Michigan football coach Lloyd Carr, died at age 5 in 2015 after being diagnosed with DIPG 14 months earlier.
In their review, researchers found a crucial factor that could influence outcomes: whether tumors with this histone mutation had invaded the surrounding brain.
Tumors from 21 patients stood out because the tumors had not invaded into the surrounding brain tissue. These patients, compared to those with more invasive tumors, had approximately four to five times longer survival rates, according to research published in the journal Acta Neuropathologica.
“These findings show that the extent of invasion into the surrounding brain tissue is really important in determining prognosis in DIPGs and similar tumors with histone mutations,” says Sriram Venneti, MD, PhD, a Michigan Medicine pathologist and the study’s senior author.
“Current guidelines lump all tumors with this type of histone mutation into the same category when it comes to prognosis,” Venneti says. “But our study suggests that we may need to consider how the tumor invades into surrounding regions of the brain. While the majority of these patients have invasive tumors, we found that those who don’t, even though a small group of patients, have a better prognosis. This information could be meaningful to their individual treatment and outcomes.”
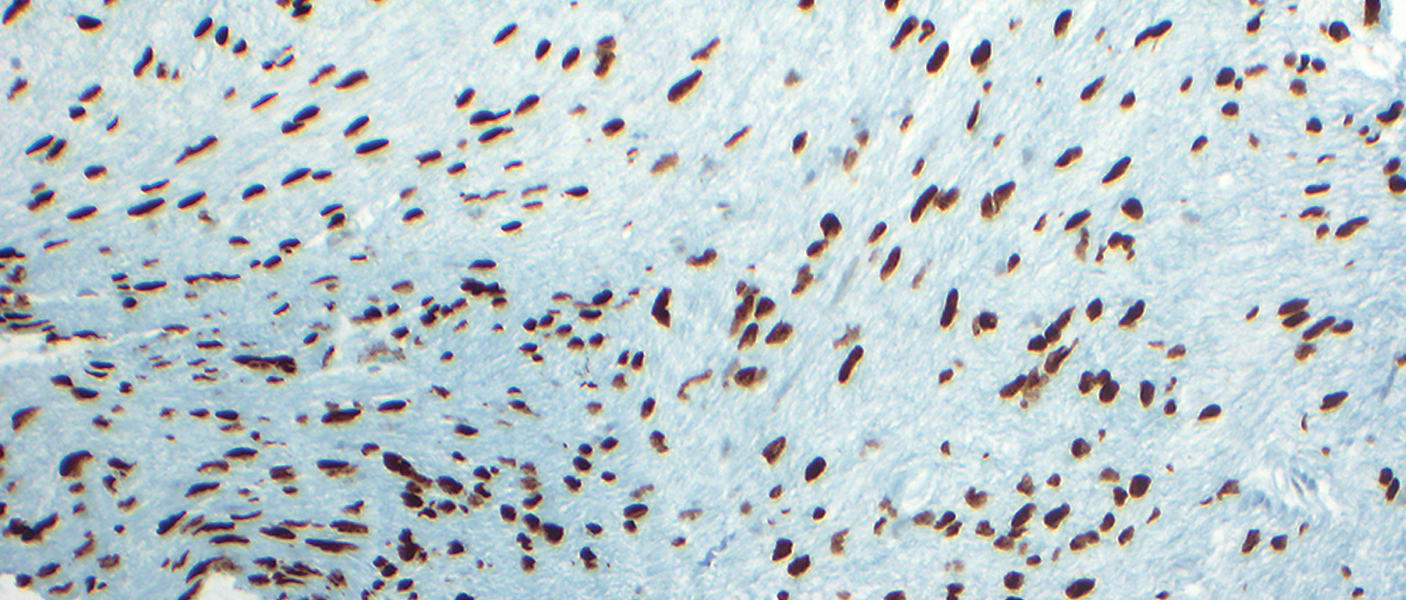
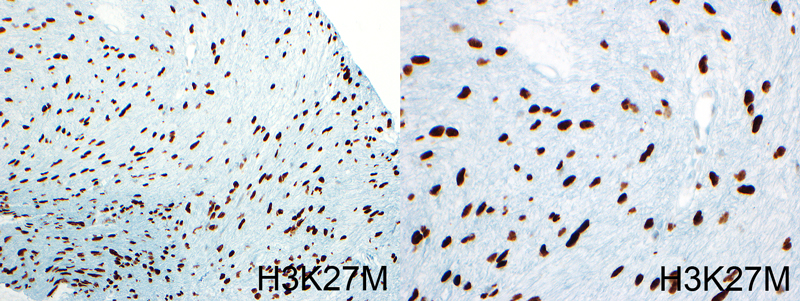 The images show are obtained from DIPG tumors stained with a marker that highlight nuclei within tumor cells (brown color). These mutations are in a protein termed histone H3 that are important for DNA packaging in the nucleus. The type of mutation is termed H3K27M and is observed in more than 80% of DIPG tumors.The research is among a series of studies expected to be published through the year that use sequencing data from pediatric brain tumors. Chad Carr’s family, along with others treated at U-M’s C.S. Mott Children’s Hospital, donated tumor tissue to DIPG research at Michigan.
The images show are obtained from DIPG tumors stained with a marker that highlight nuclei within tumor cells (brown color). These mutations are in a protein termed histone H3 that are important for DNA packaging in the nucleus. The type of mutation is termed H3K27M and is observed in more than 80% of DIPG tumors.The research is among a series of studies expected to be published through the year that use sequencing data from pediatric brain tumors. Chad Carr’s family, along with others treated at U-M’s C.S. Mott Children’s Hospital, donated tumor tissue to DIPG research at Michigan.
“Chad’s tumor, along with other DIPGs, is helping us better understand how the tumor behaves and grows and why it is so resistant against treatment,” Venneti says.
The study, a collaboration among Mott, U-M’s pathology department, the Michigan Comprehensive Cancer Center and the Mayo Clinic, comes on the heels of a recent paper specifically focused on Chad’s tumor uncovering genetic clues.
Because DIPG and tumors that bear this type of mutation are so rare and difficult to biopsy, clinical guidelines are based on relatively small patient cohorts, Venneti says. That’s what makes the size and scope of the latest study so impactful.
“We’ve never seen a statistical significance between invasive and noninvasive tumors that bear this type of histone mutation because the patient sample size was so small,” Venneti says. “By pooling together such a large number of global cases, we were able to uncover a significant but often overlooked factor. This study gives us statistical power to help answer the question of whether certain tumors should in fact be classified and treated differently.
“It is still far too early to know how this finding may guide therapy, but it is valuable for this group of patients,” he adds. “With such a rare type of tumor, research on every single one helps us get closer to finding a way to fight it.”
 ON THE COVER
ON THE COVER
Breast team reviewing a patient's slide. (From left to right) Ghassan Allo, Fellow; Laura Walters, Clinical Lecturer; Celina Kleer, Professor. See Article 2014Department Chair |

newsletter
INSIDE PATHOLOGYAbout Our NewsletterInside Pathology is an newsletter published by the Chairman's Office to bring news and updates from inside the department's research and to become familiar with those leading it. It is our hope that those who read it will enjoy hearing about those new and familiar, and perhaps help in furthering our research. CONTENTS
|
 ON THE COVER
ON THE COVER
Autopsy Technician draws blood while working in the Wayne County morgue. See Article 2016Department Chair |

newsletter
INSIDE PATHOLOGYAbout Our NewsletterInside Pathology is an newsletter published by the Chairman's Office to bring news and updates from inside the department's research and to become familiar with those leading it. It is our hope that those who read it will enjoy hearing about those new and familiar, and perhaps help in furthering our research. CONTENTS
|
 ON THE COVER
ON THE COVER
Dr. Sriram Venneti, MD, PhD and Postdoctoral Fellow, Chan Chung, PhD investigate pediatric brain cancer. See Article 2017Department Chair |

newsletter
INSIDE PATHOLOGYAbout Our NewsletterInside Pathology is an newsletter published by the Chairman's Office to bring news and updates from inside the department's research and to become familiar with those leading it. It is our hope that those who read it will enjoy hearing about those new and familiar, and perhaps help in furthering our research. CONTENTS
|
 ON THE COVER
ON THE COVER
Director of the Neuropathology Fellowship, Dr. Sandra Camelo-Piragua serves on the Patient and Family Advisory Council. 2018Department Chair |

newsletter
INSIDE PATHOLOGYAbout Our NewsletterInside Pathology is an newsletter published by the Chairman's Office to bring news and updates from inside the department's research and to become familiar with those leading it. It is our hope that those who read it will enjoy hearing about those new and familiar, and perhaps help in furthering our research. CONTENTS
|
 ON THE COVER
ON THE COVER
Residents Ashley Bradt (left) and William Perry work at a multi-headed scope in our new facility. 2019Department Chair |

newsletter
INSIDE PATHOLOGYAbout Our NewsletterInside Pathology is an newsletter published by the Chairman's Office to bring news and updates from inside the department's research and to become familiar with those leading it. It is our hope that those who read it will enjoy hearing about those new and familiar, and perhaps help in furthering our research. CONTENTS
|
 ON THE COVER
ON THE COVER
Dr. Kristine Konopka (right) instructing residents while using a multi-headed microscope. 2020Department Chair |

newsletter
INSIDE PATHOLOGYAbout Our NewsletterInside Pathology is an newsletter published by the Chairman's Office to bring news and updates from inside the department's research and to become familiar with those leading it. It is our hope that those who read it will enjoy hearing about those new and familiar, and perhaps help in furthering our research. CONTENTS
|
 ON THE COVER
ON THE COVER
Patient specimens poised for COVID-19 PCR testing. 2021Department Chair |

newsletter
INSIDE PATHOLOGYAbout Our NewsletterInside Pathology is an newsletter published by the Chairman's Office to bring news and updates from inside the department's research and to become familiar with those leading it. It is our hope that those who read it will enjoy hearing about those new and familiar, and perhaps help in furthering our research. CONTENTS
|
 ON THE COVER
ON THE COVER
Dr. Pantanowitz demonstrates using machine learning in analyzing slides. 2022Department Chair |

newsletter
INSIDE PATHOLOGYAbout Our NewsletterInside Pathology is an newsletter published by the Chairman's Office to bring news and updates from inside the department's research and to become familiar with those leading it. It is our hope that those who read it will enjoy hearing about those new and familiar, and perhaps help in furthering our research. CONTENTS
|
 ON THE COVER
ON THE COVER
(Left to Right) Drs. Angela Wu, Laura Lamps, and Maria Westerhoff. 2023Department Chair |

newsletter
INSIDE PATHOLOGYAbout Our NewsletterInside Pathology is an newsletter published by the Chairman's Office to bring news and updates from inside the department's research and to become familiar with those leading it. It is our hope that those who read it will enjoy hearing about those new and familiar, and perhaps help in furthering our research. CONTENTS
|
 ON THE COVER
ON THE COVER
Illustration representing the various machines and processing used within our labs. 2024Department Chair |

newsletter
INSIDE PATHOLOGYAbout Our NewsletterInside Pathology is an newsletter published by the Chairman's Office to bring news and updates from inside the department's research and to become familiar with those leading it. It is our hope that those who read it will enjoy hearing about those new and familiar, and perhaps help in furthering our research. CONTENTS
|
 ON THE COVER
ON THE COVER
Rendering of the D. Dan and Betty Khn Health Care Pavilion. Credit: HOK 2025Department Chair |

newsletter
INSIDE PATHOLOGYAbout Our NewsletterInside Pathology is an newsletter published by the Chairman's Office to bring news and updates from inside the department's research and to become familiar with those leading it. It is our hope that those who read it will enjoy hearing about those new and familiar, and perhaps help in furthering our research. CONTENTS
|

MLabs, established in 1985, functions as a portal to provide pathologists, hospitals. and other reference laboratories access to the faculty, staff and laboratories of the University of Michigan Health System’s Department of Pathology. MLabs is a recognized leader for advanced molecular diagnostic testing, helpful consultants and exceptional customer service.